ISO 17025 2017 CHANGES ISO
Some Support Processes: Though ISO 17025:2017 includes many support and resource requirements as it applies to calibration laboratories, but it does not include all the requirements from ISO 9001:2015.Monitoring and Measurement: While there are some requirements of monitoring and measurement in ISO 17025:2017, these do not address all of the requirements of ISO 9001:2015.Leadership: Although some leadership commitment is there, there is no listing of leadership commitments, including the quality policy, in ISO 17025:2017.Quality Objectives: ISO 9001:2015 has important improvement goals for the QMS which are not included in ISO 17025:2017.Context of The Organization: ISO 17025 does not talk about identifying and specifying your interested parties, internal and external issues or about establishing the scope of our QMS.If you fulfill just the minimum requirements of ISO 17025:2017 instead of requirements of ISO 9001:2015, some of the things that will be missing from your QMS are: However, it is important to note that while ISO 9001 implementation is nice to have, it is not required to comply with ISO 17025:2017. On the other hand, Option B states that if you have a QMS which meets the requirements of ISO 9001:2015, then you have everything that you need. If the laboratory has not implemented ISO 9001, then a minimum set of QMS requirements which are needed for the applicability of ISO 17025:2017, need to me fulfilled.
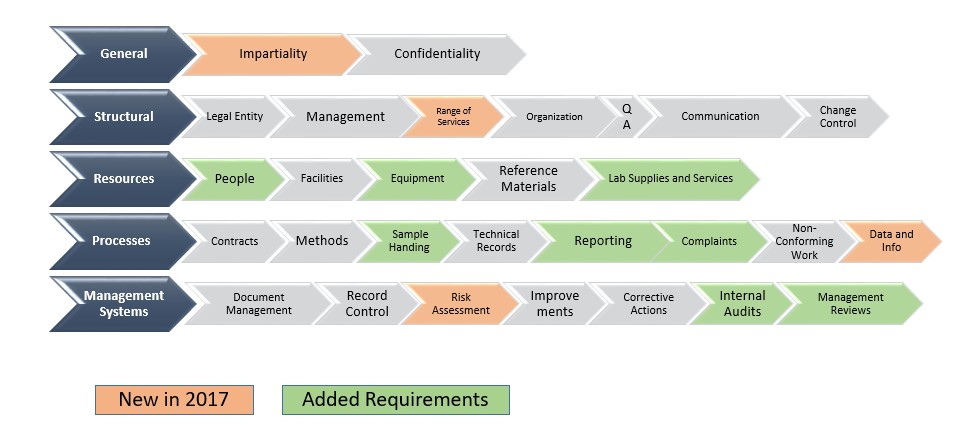
ISO 17025:2017 explains that a Quality Management System is necessary for the proper functioning of a calibration laboratory, and then provides you with two options i.e. ISO 9001:2015 can be applied to all types of industries as its requirements are very generic, whereas ISO 17025:2017 is applicable only on calibration laboratories as its requirements are very specific as to what you need to implement in calibration laboratories. The difference between ISO 17025:2017 and ISO 9001:2015 is in the applicability of the standards. Drop an e-mail at or call at +91-9316744482 to know more about this training. conducts ISO 17025:2017 Upgradation and Internal Auditor Course at reasonable fees. Addition of all laboratory activities in the scope of the standard which includes calibration, testing and sampling related with subsequent testing and calibration.Įurotech Assessment and Certification Services Pvt.Recognition and incorporation of computer systems, e-records, and production of electronic reports and results.Increased flexibility in the guidelines for documented information, procedures, organizational responsibilities and processes.A new topic on risk based thinking has been added which has enabled reduction in standard’s prescriptive guidelines and replaced them with performance based guidelines.This updated standard is based on a new structure which aligns it with other existing ISO/IEC conformity assessment standards.
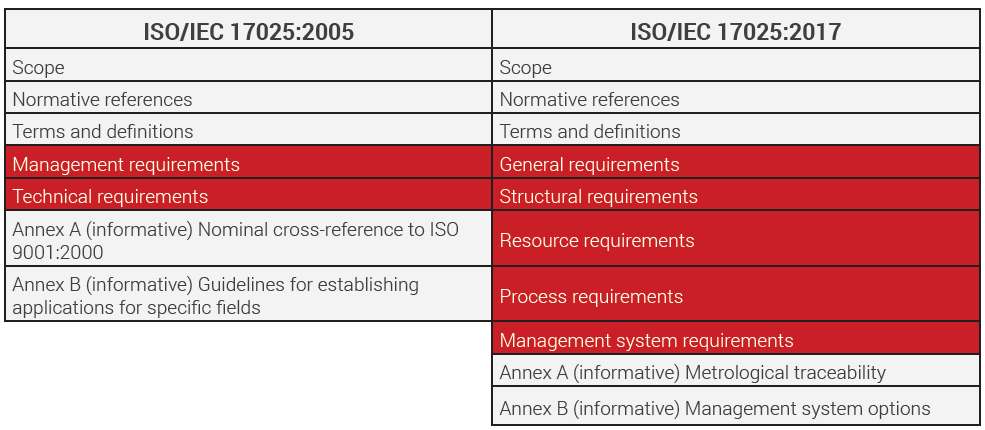
To meet these changes and to establish a harmonization among other existing international standards, ISO 17025:2005 was revised and upgraded to ISO 17025:2017.

Since then, the market has undergone many changes. The older version of ISO 17025 was the second edition which was published in 2005.
